Linked Michaelis-Menten type models for
methanogenesis data
“A stable isotope titration method for measurement of
the contribution of acetate and carbon dioxide reduction to methane production”
By N.D. Gray, J.N.S. Matthews and I.M. Head
MMLINK:
a program to fit the linked Michaelis-Menten type equations in this report
How to get
starting values
As described in the document
on statistical methods, the parameters
of the various models are estimates by choosing those values which maximise the
likelihood of the data, given the model.
For each model the log-likelihood is maximised analytically over all but
two of the parameters in the model, namely B and K. The maximising values for these parameters
are found by numerical methods. These methods require the user to enter
suitable initial values – educated guesses – about the values of the parameters
and the software successively improves on these until the maximizing values are
found. This page gives the user some
guidance on how to make these educated guesses.
The analysis to obtain
plausible starting values uses only the data on 13CH4. According to the analysis, it is the rate of
production of 13CH4 which follows the standard
Michaelis-Menten equation, given in the first equation in the document on statistical methods. For large values of [Slabelled],
the rate of production of 13CH4 approaches B. The other parameter, K, is the value
of [Slabelled] when the rate of methane production is half of B. Consequently, if the user plots the rates of
production of 13CH4 (y value) against [Slabelled]
(x value) then the saturation level of the production rate and the
substrate concentration at which the rate of production is half the saturation
value can be used as starting values for the numerical method.
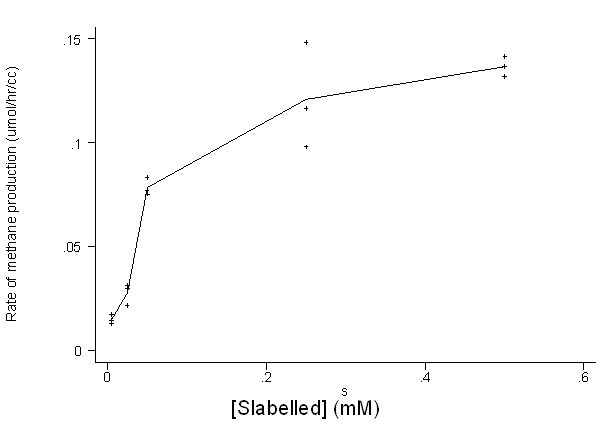
For the data used in the
above report, this plot is shown below. Individual
observations are plotted and the mean production rates at each distinct substrate
concentration are joined by straight lines.
The saturation level is plausibly 0.15 mmol/hr/cc and the substrate concentration when the
production rate is 0.075 mmol/hr/cc is reasonably taken to be 0.05 mM.
Alternative methods for
getting starting values are possible.
Various transformations of the Michaelis-Menten equation can be made to
allow B and K to be estimated through simple linear regression on
suitably transformed variables. We have
adopted the above approach because it is a good discipline to plot the data. Once this is done, starting values of
adequate accuracy can easily be read off the plot.